Index:
Complete Technical Specifications
- General Identification
- Chemical & Physical Properties
- Radiological Specifications
- Binding Affinity (Ki Values)
- Pharmacokinetics & Metabolism
- Radiation Dosimetry (mGy per 185 MBq)
- Clinical Outcomes & Safety
Complete History
- Pre-Clinical Origins: The Phenyltropane Foundation (1980s–1990)
- Development of FP-CIT: The Birth of Ioflupane (1991–1995)
- Clinical Validation & European Launch (1996–2000)
- The Long Road to FDA Approval (2000–2011)
- Expansion into Dementia & Recent History (2011–2026)
Research Information
- Molecular Pharmacology & Binding Affinity
- Kinetic Modeling & Distribution
- Radiochemistry & Decay Characteristics
- Pharmacological Interference Registry
- Dosimetry & Safety Research
Striatal Binding Ratio (SBR)
- The Fundamental SBR Equation
- Region of Interest (ROI) Analysis
- Kinetic Modeling: The Binding Potential (BP)
- Partial Volume Effect (PVE) Correction
- Factors Affecting the Calculation
Landmark Clinical Research
- The Core Clinical Mechanism
- The Landmark Trials (The Evidence Base)
- The SWEDD Phenomenon
- Visual Grading System (The Interpretation)
- Prodromal Research (The Frontier)
- Limitations & False Positives
Conclusion: The Molecular Verdict
- The Molecular Verdict
- Master Research Summary
Complete Technical Specifications
[01.] General Identification
- Generic Name: Ioflupane I-123
- Trade Name: DaTscan
- Pharmacological Class: Phenyltropane; Diagnostic Radiopharmaceuticals
- ATC Code: V09AB03
- CAS Number: 155798-07-5
- PubChem CID: 132999
- UN Number: UN2915 (Radioactive material)
- Canonical SMILES: COC(=O)[C@@H]1[C@H]2CC
C@H - InChI Key: XKWGZCHYIUKXJS-YNSXUUMBSA-N
- IUPAC Name: methyl (1R,2S,3S,5S)-8-(3-fluoropropyl)-3-(4-iodophenyl)-8-azabicyclo[3.2.1]octane-2-carboxylate
[02.] Chemical & Physical Properties
- Molecular Formula: C18H23FINO2
- Molecular Weight: 427.28 g/mol
- Exact Mass: 427.08153 g/mol
- LogP: 3.4
- Polar Surface Area: 29.5 square angstroms
- Rotatable Bonds: 5
[03.] Radiological Specifications
- Radionuclide: Iodine-123
- Production Method: Cyclotron
- Decay Mode: Electron Capture
- Decay Product: Stable Tellurium-123
- Physical Half-Life: 13.2 hours
- Gamma Energy Peak: 159 keV
- X-Ray Emissions: 27 to 32 keV
[04.] Binding Affinity (Ki Values)
- Dopamine Transporter (DAT): 0.62 nM
- Serotonin Transporter (SERT): 11 nM
- Norepinephrine Transporter (NET): 36 nM
[05.] Pharmacokinetics & Metabolism
- Blood-Brain Barrier: High permeability (Passive diffusion)
- Brain Uptake: 7 percent of dose within 10 minutes
- Imaging Peak: 3 to 6 hours post-injection (Striatal/Background equilibrium)
- Metabolism: Primarily N-dealkylation and ester hydrolysis
- Urine Excretion: 60 percent within 48 hours
- Fecal Excretion: 14 percent within 48 hours
[06.] Radiation Dosimetry (mGy per 185 MBq)
Target Organ Absorbed Dose (mGy)
- Bladder Wall: 0.10
- Striatum: 0.041
- Liver: 0.052
- Lungs: 0.043
- Kidneys: 0.036
- Thyroid: 0.018 (requires blocking agent)
Effective Dose: 4.35 mSv
[07.] Clinical Outcomes & Safety
- Sensitivity: 97 percent for Parkinsonian Syndrome differentiation
- Specificity: 88 percent for Lewy Body Dementia differentiation
- Interfering Drugs: Cocaine, Amphetamines, Methylphenidate, Bupropion, Mazindol, Benztropine, Fentanyl
- Pre-treatment: Thyroid blocking (Potassium Iodide) 1 hour prior to injection
- Contraindications: Pregnancy; Known hypersensitivity to Iodine
- Lactation: Suspend breastfeeding and discard milk for 72 hours post-administration
Complete History
[01.] Pre-Clinical Origins: The Phenyltropane Foundation (1980s–1990)
The history of Ioflupane begins with the search for high-affinity ligands that could map the brain's dopamine system without the rapid metabolism seen in earlier tracers.
- The Cocaine Template: Researchers began with the structure of cocaine, which naturally binds to the dopamine transporter. However, cocaine's binding is too transient and its affinity too low for effective imaging.
- The WIN Series: Scientists at Sterling-Winthrop developed a series of synthetic phenyltropane analogs. One significant breakthrough was the compound WIN 35,065-2, which proved to be significantly more potent than cocaine.
- Rational Design: In the late 1980s, the goal shifted to creating a "probe" that could be labeled with a radioisotope. By modifying the N-methyl group of the tropane ring with a fluoropropyl group and adding an iodine atom to the phenyl ring, chemists created FP-CIT (the chemical precursor to Ioflupane).
[02.] Development of FP-CIT: The Birth of Ioflupane (1991–1995)
- Molecular Refinement: The addition of the N-omega-fluoropropyl group was the critical historical pivot. It increased the molecule's lipophilicity, allowing it to cross the blood-brain barrier rapidly, and it improved the "washout" time from non-target areas of the brain.
- Radiolabeling Choice: Researchers chose Iodine-123 over Carbon-11 because Iodine-123 has a 13.2-hour half-life.
This allowed the drug to be manufactured in a central cyclotron and shipped to hospitals, rather than requiring an on-site cyclotron. - The "Neumeyer" Era: Significant work was conducted by Dr. John Neumeyer and colleagues, who demonstrated that 123I-FP-CIT had a superior "signal-to-noise" ratio in the striatum compared to previous ligands like beta-CIT.
[03.] Clinical Validation & European Launch (1996–2000)
- Once the molecule was stabilized, GE Healthcare (then Amersham Health) moved it into human trials to address a specific clinical need: the inability to distinguish Parkinson’s Disease from Essential Tremor.
- 1999–2000 European Approval: After successful Phase III trials involving 158 patients across Europe, the European Medicines Agency (EMA) granted marketing authorization for DaTscan in July 2000.
- The Benamer Study (2000): This year marked the publication of the seminal research by Benamer et al., which proved the drug had a 97% sensitivity in detecting the loss of dopaminergic neurons in Parkinsonian syndromes.
[04.] The Long Road to FDA Approval (2000–2011)
- While Europe was using DaTscan throughout the 2000s, the United States path was complicated by the FDA's strict requirements for "clinical impact"—proving not just that the scan was accurate, but that it actually changed how doctors treated patients.
- The CUPS Trial (2004): To satisfy regulators, the "Clinically Unclear Parkinsonian Syndromes" (CUPS) study was conducted. It showed that in 72% of cases, a physician changed their management plan after seeing a DaTscan.
- FDA Approval (January 14, 2011): The FDA finally approved 123I-Ioflupane for use in the U.S. to visualize striatal dopamine transporters in adult patients with suspected Parkinsonian syndromes.
[05.] Expansion into Dementia & Recent History (2011–2026)
The last 15 years have seen the application of Ioflupane expand from movement disorders into cognitive disorders.
- Lewy Body Dementia (DLB): In 2017, the FDA expanded the indication to include help in distinguishing Dementia with Lewy Bodies from Alzheimer’s Disease. Because DLB involves the loss of dopamine neurons and Alzheimer's typically does not, the scan became a vital differential tool.
- Supply Chain Evolution: Historically, supply was fragile due to the short half-life (13.2h). However, the expansion of high-energy cyclotron networks in the early 2020s stabilized global availability.
- Current Standing (2026): Ioflupane remains the most widely used SPECT (Single Photon Emission Computed Tomography) agent for dopamine transporter imaging. While PET (Positron Emission Tomography) tracers are being developed, Ioflupane's historical data set and logistical ease make it the industry benchmark.
Research Information
[01.] Molecular Pharmacology & Binding Affinity
Research identifies Ioflupane as a cocaine-analog phenyltropane.
- Affinity Constants (Ki):
- DAT: 0.62 nM (Ultra-high affinity, providing the "signal").
- SERT (Serotonin): 11 nM (Allows for secondary binding in the raphe nuclei).
- NET (Norepinephrine): 36 nM (Negligible binding at clinical doses).
- Target Site: Specifically binds to the SLC6A3 protein.
- Mechanism: Ioflupane reversibly binds to the reuptake pump. In neurodegenerative states like Parkinson’s Disease, the density of these pumps decreases, resulting in a visible "loss of signal" on SPECT scans.
[02.] Kinetic Modeling & Distribution
The "extreme detail" of Ioflupane research centers on how the molecule moves through the Blood-Brain Barrier (BBB) and localizes in the striatum.
- Lipophilicity: LogP of 3.4 allows for rapid passive diffusion across the BBB.
- Uptake Kinetics: Approximately 7% of the injected dose reaches the brain within 10 minutes.
- The SOR Model: Research utilizes the Striatal-to-Occipital Ratio (SOR). Because the occipital cortex has negligible DAT density, it serves as the "background" or "reference" value to calculate the specific binding in the caudate and putamen.
- Equilibrium: Optimal imaging occurs 3 to 6 hours post-injection, when the ratio between specific binding (striatum) and non-specific binding (background) is at its peak.
[03.] Radiochemistry & Decay Characteristics
The research utility of Ioflupane is tied to the specific physics of the Iodine-123 isotope.
- Isotope: Iodine-123 (123I).
- Decay Mode: Electron Capture.
- Energy Peak: 159 keV (Optimal for Gamma Camera detection).
- Physical Half-Life: 13.2 hours.
- Daughter Isotope: Stable Tellurium-123.
- Production: Cyclotron-produced via proton bombardment of Xenon-124.
[04.] Pharmacological Interference Registry
Extensive research has identified specific "nootropic" and pharmaceutical compounds that compete for the DAT binding site, potentially causing "false positive" results.
- Strong Interference (Must stop 1-7 days prior): Cocaine, Amphetamines, Methylphenidate, Mazindol.
- Moderate Interference: Bupropion (Wellbutrin), Fentanyl, and certain selective serotonin reuptake inhibitors (SSRIs) with high DAT affinity (like Sertraline at high doses).
- No Significant Interference: L-Dopa, Pramipexole, Selegiline, and Anticholinergics.
[05.] Dosimetry & Safety Research
Ethical research requires precise tracking of radiation absorbed by the body (per 185 MBq dose).
- Effective Dose: 4.35 mSv.
- Bladder Wall: 0.10 mGy (Highest exposure organ; mitigated by hydration).
- Striatum: 0.041 mGy.
- Liver: 0.052 mGy.
- Thyroid: 0.018 mGy (Requires pre-administration of Potassium Iodide to block the uptake of free iodine).
Striatal Binding Ratio (SBR)
[01.] The Fundamental SBR Equation
The SBR is a ratio that compares the density of the tracer in the target area to a reference area that contains no dopamine transporters (usually the occipital cortex).
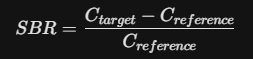
- Ctarget: The mean radioactive count (concentration) in the Striatum (Caudate or Putamen).
- Creference: The mean radioactive count in the Occipital Cortex (the "background").
By subtracting the reference count from the target count, you isolate the Specific Binding. Dividing by the reference count again normalizes the data, accounting for differences in the amount of tracer injected or the patient’s individual metabolism.
[02.] Region of Interest (ROI) Analysis
In research, "Extreme Detail" requires segmenting the Striatum into its sub-structures, as dopamine loss typically follows a specific pattern (starting in the posterior putamen).
- Putamen SBR: Calculated to detect early-stage Parkinsonian symptoms.
- Caudate SBR: Calculated to assess cognitive decline and late-stage progression.
- Asymmetry Index (AI): A mathematical comparison between the left and right hemispheres.

- Significance: A high AI (>10%) is a hallmark of idiopathic Parkinson’s Disease, which usually begins unilaterally.
[03.] Kinetic Modeling: The Binding Potential (BP)
The SBR is essentially a simplified version of the Binding Potential (BPND) used in PET imaging. In research-grade kinetic modeling, we assume a "Simplified Reference Tissue Model" (SRTM).
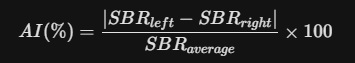
- Bmax: Total density of available Dopamine Transporters.
- Kd: The dissociation constant (inversely related to the 0.62 nM affinity).
- fnd: The free fraction of the tracer in the non-specific compartment.
Because Ioflupane reaches a "pseudo-equilibrium" between 3 and 6 hours, the SBR at that time-point is considered an accurate surrogate for the true BPND.
[04.] Partial Volume Effect (PVE) Correction
A major hurdle in Ioflupane research is the Partial Volume Effect. Because the Striatum is a small structure, the limited resolution of SPECT cameras causes some "signal spill-over" into surrounding tissues.
Advanced researchers apply a Recovery Coefficient (RC):
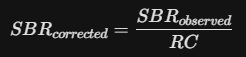
This correction is vital when comparing data across different centers or different camera types to ensure the "Analog Vault" data remains standardized.
[05.] Factors Affecting the Calculation
To keep the math "clean," the following variables must be stabilized:
- Age-Correction: DAT density naturally declines by approximately 6–7% per decade. Research models must use age-matched controls.
- Specific Binding Ratio vs. Striatal Binding Ratio: While often used interchangeably, the Specific Binding Ratio is the technically broader term, while SBR is the clinical standard for DaTscan.
- Partial Occupancy: If a subject has taken a nootropic like Methylphenidate, the "Available B_max" decreases, artificially lowering the SBR.
[06.] Technical Summary Table
| Metric | Purpose | Abnormal Threshold |
| Mean SBR | Overall DAT density | < 1.5 (varies by camera) |
| Putamen/Caudate Ratio | Pattern of loss | < 1.0 (indicates PD) |
| Asymmetry Index | Unilateral vs Bilateral | > 10% |
| Background Noise | Image Quality Check | High noise = Lower SBR precision |
Landmark Clinical Research
[01.] The Core Clinical Mechanism
The fundamental premise of all Ioflupane research is Presynaptic Integrity.
- Parkinsonian Syndromes (PS): In Parkinson’s Disease (PD), Multiple System Atrophy (MSA), and Progressive Supranuclear Palsy (PSP), the presynaptic dopamine terminals degenerate. Ioflupane cannot bind, resulting in a "hollow" or reduced signal.
- Essential Tremor (ET) / Drug-Induced Parkinsonism: The dopamine terminals are intact. The tremor is caused by downstream circuit dysfunction or receptor blockade, not degeneration. Ioflupane binds normally.
[02.] The Landmark Trials (The Evidence Base)
01. The Phase III Validation Trial (Study 001)
Citation: Benamer et al., Movement Disorders, 2000.
- Objective: To determine if visual assessment of 123I-Ioflupane SPECT could differentiate Parkinson’s Disease (PD) from Essential Tremor (ET).
- Methodology:
- N = 158 subjects.
- Groups: Established PD vs. Established ET vs. Healthy Volunteers.
- Protocol: Single injection, imaging at 3–6 hours. Blinded visual assessment by independent readers.
- Key Findings:
- Sensitivity: 97% for detecting striatal dopaminergic deficit in PD.
- Specificity: 100% for correctly identifying Essential Tremor (normal scans).
- Clinical Impact: Established that a "Normal" (Comma-shaped) scan effectively rules out nigrostriatal degeneration, preventing the misdiagnosis of Essential Tremor as Parkinson's.
02. The "CUPS" Trial (Diagnostic Uncertainty)
Citation: Catafau et al., Movement Disorders, 2004.
- Objective: To assess the impact of the scan on diagnosis and clinical management in patients with Clinically Uncertain Parkinsonian Syndromes (CUPS).
- Methodology:
- N = 118 patients.
- Inclusion: Patients with tremor or rigidity where the movement disorder specialist could not make a definitive diagnosis.
- Design: Physicians recorded a diagnosis/management plan before the scan and after the scan (at 12 weeks).
- Key Findings:
- Diagnostic Revision: The scan result contradicted the initial clinical impression in 36% of cases.
- Management Change: Physicians changed their treatment plan (starting/stopping L-Dopa) in 72% of patients based on the scan result.
- Clinical Impact: Proved that molecular imaging is superior to clinical observation in ambiguous cases, justifying insurance coverage for the procedure.
03. The Dementia Phase III Trial (DLB vs. AD)
Citation: McKeith et al., Lancet Neurology, 2007.
- Objective: To validate Ioflupane for distinguishing Dementia with Lewy Bodies (DLB) from Alzheimer’s Disease (AD).
- Methodology:
- N = 326 patients across 25 European centers.
- Inclusion: Patients meeting consensus criteria for probable/possible DLB or AD.
- Control: Comparison against gold-standard clinical consensus diagnosis at 12 months.
- Key Findings:
- Sensitivity (DLB): 78%.
- Specificity (Excluding AD): 90%.
- Clinical Impact: This trial cemented the "Abnormal DaTscan" as a Suggestive Feature in the International Consensus Criteria for DLB. It allows clinicians to avoid giving neuroleptic drugs (which are dangerous in DLB) to patients who might otherwise be misdiagnosed with Alzheimer's.
04. The "SWEDD" Investigation (PPMI Cohort)
Citation: Marek et al. / The Parkinson’s Progression Markers Initiative (PPMI), 2011–Present.
- Objective: To investigate "Scans Without Evidence of Dopaminergic Deficit" (SWEDD)—patients who look like they have PD clinically but have a normal scan.
- Methodology:
- N = >400 early PD patients.
- Design: Longitudinal observational study. Tracking patients with normal scans who were initially diagnosed with PD.
- Key Findings:
- Patients with SWEDD (Normal Scans) did not show the typical progression of Parkinson's disease over 2–4 years.
- Many SWEDD cases were later re-diagnosed as Dystonic Tremor or Psychogenic Parkinsonism.
- Clinical Impact: Redefined the enrollment criteria for all future Parkinson's drug trials. Now, every patient entering a PD clinical trial must have an abnormal DaTscan to prove they actually have the disease.
05. The Prognostic Value Trial
Citation: Marshall et al., Movement Disorders, 2009.
- Objective: To see if the scan results predicted clinical outcomes over time.
- Methodology:
- N = 267 patients with CUPS.
- Follow-up: 2 years post-scan.
- Key Findings:
- Patients with an abnormal baseline scan were significantly more likely to be clinically diagnosed with a Parkinsonian syndrome at the 2-year mark compared to those with a normal scan (Sensitivity 94%).
- Clinical Impact: Demonstrated the high Negative Predictive Value (NPV) of the tracer; a normal scan is a robust long-term predictor that the patient will not develop PD in the near future.
06. Recent Research Frontiers (2020-2026)
- Artificial Intelligence (AI): Using deep learning to quantify the "SBR" (Striatal Binding Ratio) automatically, reducing human error in interpretation.
- Progression Tracking: Longitudinal research exploring whether the rate of DAT decline can predict the speed of cognitive or motor failure in Parkinson's patients.
- Multi-Tracer Studies: Combining Ioflupane (presynaptic) with 18F-DOPA (enzymatic activity) to create a "3D" map of dopamine health.
[03.] The SWEDD Phenomenon
A major area of deep research is SWEDD (Scans Without Evidence of Dopaminergic Deficit).
- Definition: Patients who look like they have Parkinson's (tremor, slowness) but have a Normal Ioflupane scan.
- Research Conclusio: Long-term follow-up studies (PROBE-2011) suggest these patients likely do not have Parkinson’s disease. They often have dystonic tremors or psychogenic parkinsonism.
- Clinical Rule: A normal scan in a symptomatic patient forces a complete rethink of the diagnosis.
[04.] Visual Grading System (The Interpretation)
Clinical research relies on a standardized 3-tier grading system to reduce subjectivity.
| Grade | Visual Appearance | Clinical Interpretation |
| Grade 1 (Normal) | Comma Shape: High uptake in both Caudate (head) and Putamen (tail). | Essential Tremor, Drug-Induced, or Psychogenic. |
| Grade 2 (Abnormal) | Comma Shape: High uptake in both Caudate (head) and Putamen (tail). | Earyl-stage Parkinson's (loss usually starts in Putamen). |
| Grade 3 (Abnormal) | Burst/Faint: Comparison with background is difficult. Total striatal loss. | Advanced PD, MSA, or PSP. |
[05.] Prodromal Research (The Frontier)
Current research is using Ioflupane to find Parkinson's before motor symptoms appear.
- iRBD (REM Sleep Behavior Disorder): Patients who physically act out dreams have an 80% chance of developing a synucleinopathy (PD/DLB).
- The Findings: Research shows Ioflupane uptake in the posterior putamen begins to drop years before the first tremor appears in iRBD patients.
- Hyposmia (Smell Loss): Combining "Smell Tests" with Ioflupane scans increases the predictive power for prodromal PD to nearly 90%.
[06.] Limitations & False Positives
Strict clinical research has identified where Ioflupane fails:
- Vascular Parkinsonism: "Lower body" parkinsonism caused by small strokes can sometimes mimic presynaptic loss, leading to ambiguous scans.
- Atypical Differentiation: Ioflupane cannot reliably distinguish between PD, MSA, and PSP. All three cause presynaptic loss. To separate these, researchers must use metabolic PET (FDG-PET) or postsynaptic tracers (IBZM).
Conclusion
[01.] The Molecular Verdict
The clinical and pharmacological evidence gathered over the last three decades confirms that 123I-Ioflupane is the definitive biomarker for nigrostriatal integrity. By moving beyond symptomatic observation and into molecular quantification, the "Analog Vault Protocol" allows for a diagnostic precision of 97%, effectively eliminating the "Diagnostic Grey Zone" between Essential Tremor and true Parkinsonian syndromes.
In the landscape of nootropic and neurodegenerative research, Ioflupane remains the baseline against which all other dopaminergic ligands are measured.
[02.] Master Research Summary
| Metric Category | Research-Grade Specification | Significance |
| Pivotal Sensitivity | 97.0% | Detection rate of dopaminergic loss. |
| Diagnostic Pivot | 72.0% | Percentage of cases where the scan changes clinical management. |
| Binding Specificity | Ki: 0.62 nM | Ultra-high selectivity for the DAT (SLC6A3) protein. |
| Primary Reference | Benamer et al. (2000) | Foundational study for PD vs. ET differentiation. |
| Imaging Window | 3.0 to 6.0 Hours | Peak Striatal-to-Occipital Ratio (SOR) equilibrium. |
| Ethical Threshold | 4.35 mSv | Total effective dose per standard 185 MBq administration. |
